Turkovac Clinical Trials Booster Phase II
TSB-VAC-COV-TUR-RF2.02
"Double-Blind, Randomized, Controlled, Multi-Centre Phase 2 Clinical Study to Determine the Safety, Efficacy, and Immunogenicity of Booster Vaccination Against SARS-CoV-2 (Hybrid COV-Booster TR)"
The aim of this study, the primary objectives are to determine the safety of booster vaccine doses against SARS-CoV-2 following at least 90 and at most 270 days after the 2nd dose of a homologous 2-dose primary regimen. The secondary objective is to determine the immunogenicity of booster vaccine doses against SARS-CoV-2 following at least 90 and at most 270 days after the 2nd dose of a homologous 2-dose primary regimen. The study, which began with the recruitment of the first volunteer on 12.07.2021, was completed one year after the recruitment of the last volunteer on 12.07.2021. In total, 236 volunteers were screened, and 222 volunteers were vaccinated. In the Hybrid COV-Booster TR study, which aims to determine the safety and immunogenicity of booster doses of the inactivated COVID-19 vaccine administered to individuals for whom at least 90 days and at most 270 days have passed after the 2nd dose of the CoronaVac vaccine, all 222 volunteers included in the study completed the study. Consequently, when looking at the relevant data belonging to the TURKOVAC and CoronaVac arms in terms of safety, no findings threatening safety have been encountered in either group. None of the systemic and local adverse events created clinical suspicion and did not negatively affect the study. In light of these data, it has been observed that the booster doses of the inactivated COVID-19 vaccines used within the scope of the study are safe. In terms of the efficacy of booster dose vaccination, no significant difference was observed between the TURKOVAC and the comparator product CoronaVac arms. Both vaccines administered as a booster dose led to an increase in antibody titres with an acceptable safety profile.
"Double-Blind, Randomized, Controlled, Multi-Centre Phase 2 Clinical Study to Determine the Safety, Efficacy, and Immunogenicity of Booster Vaccination Against SARS-CoV-2"
TSB-VAC-COV-TUR-RF2.02
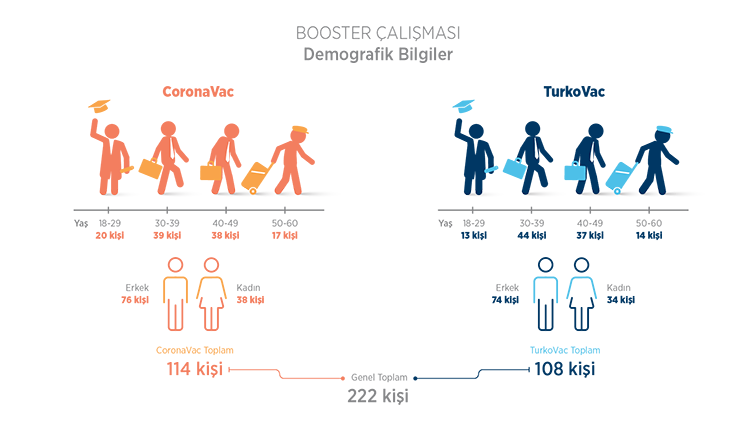
A total of 222 individuals between the ages of 18–60 were included in this study, in which the efficacy and safety of administering the COVID-19 inactivated vaccine as a booster dose to individuals who had been vaccinated with the COVID-19 inactivated vaccine (CoronaVac) were evaluated. CoronaVac was administered to 114 volunteers, and the TURKOVAC vaccine was administered as a booster dose to 108 volunteers.
"Double-Blind, Randomized, Controlled, Multi-Centre Phase 2 Clinical Study to Determine the Safety, Efficacy, and Immunogenicity of Booster Vaccination Against SARS-CoV-2"
TSB-VAC-COV-TUR-RF2.02
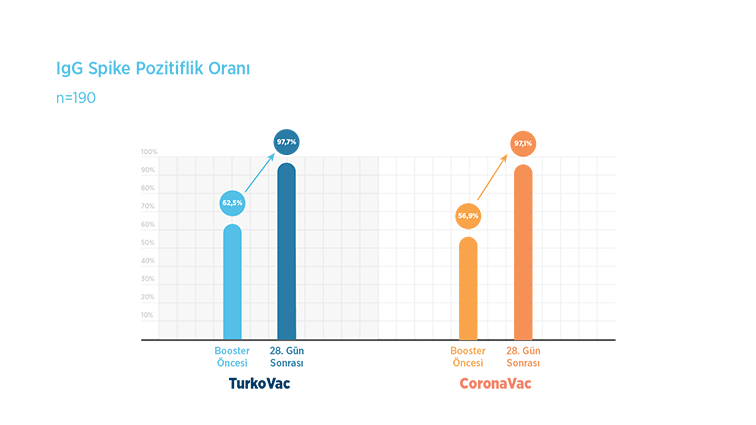
While the protective response of the previous vaccine continued in 62.5% of the volunteers who received the TURKOVAC vaccine as a booster dose before the vaccine dose, protection was achieved in 97.7% of the volunteers after the vaccine.
While the pre-vaccination protection continued at 56.9% for those who received the CoronaVac booster dose, this rate increased to 97.1% after the vaccination.
It was observed that the booster dose of both vaccines was effective.
"Double-Blind, Randomized, Controlled, Multi-Centre Phase 2 Clinical Study to Determine the Safety, Efficacy, and Immunogenicity of Booster Vaccination Against SARS-CoV-2"
TSB-VAC-COV-TUR-RF2.02
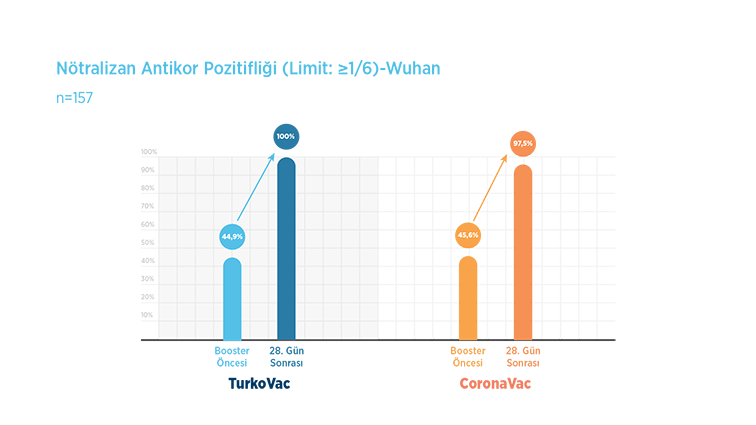
When the inhibition rate of the booster vaccine dose against the Wuhan strain, which is the original strain of SARS-CoV-2 that causes COVID-19, was examined in a laboratory environment, it was observed that while the effect of the previous vaccination continued at a rate of 44.9% before the vaccine in the TURKOVAC vaccine arm, this rate was 100% after the booster dose of the TURKOVAC vaccine. In the CoronaVac vaccine, the inhibition rate, which was 45.6% before the booster vaccine, became 97.5% after the vaccine.
With these results, it has been shown that both vaccines provide effective protection against the Wuhan strain after the booster dose.













