Turkovac Clinical Trials Primary Phase III
TSB-VAC-COV-TUR-F3.01
"A Randomized, Multi-Centre, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
The aim of this study, the primary objective is to evaluate the efficacy of two doses of TURKOVAC and two doses of CoronaVac vaccines against RT-PCR confirmed symptomatic COVID-19 disease. Secondary objectives are; to evaluate the efficacy of one dose of TURKOVAC and one dose of CoronaVac vaccines against RT-PCR confirmed symptomatic COVID-19 disease, to evaluate the safety of TURKOVAC and CoronaVac vaccines, to evaluate the immunogenicity of TURKOVAC and CoronaVac vaccines, to determine the seropositivity rate of SARS-CoV-2 specific binding antibody at 42, 56, 70, 168, and 224 days after the first dose, to evaluate SARS-CoV-2 neutralizing antibodies by neutralization test on the day of the first dose and at 42, 56, 70, 168, and 224 days after the first dose among volunteers receiving TURKOVAC and volunteers receiving CoronaVac, to evaluate T-cells (T-cell activation) specific to the SARS-CoV-2 protein peptide pool on the day of the first dose and at 42, 70, 168, and 224 days after the first dose, to evaluate the efficacy of two doses of the TURKOVAC vaccine against severe COVID-19 cases and the rates of hospitalization and death, and to determine the incidence of adverse reactions and serious adverse events. The study began with the recruitment of the first volunteer on 22.06.2021 and was completed one year after the recruitment of the last volunteer on 07.01.2022. 1,296 volunteers were screened, and 1,290 volunteers were vaccinated. The volunteers to be included in the study were randomly assigned to one of two arms: either the inactive study vaccine TURKOVAC, to be administered in 2 doses of $3 \mu g / 0.5 ml$, or the CoronaVac vaccine, to be administered in 2 doses of $3 \mu g / 0.5 ml$. In the Primary Phase 3 study, which aims to evaluate the efficacy of two doses of TURKOVAC and two doses of CoronaVac vaccines against RT-PCR confirmed symptomatic COVID-19 disease, no significant difference was observed between the TURKOVAC and the comparator product CoronaVac arms in terms of the efficacy of vaccination. In terms of safety, according to the analysis results, although it was observed that the adverse effects developing in the anatomical regions where the vaccines were administered were relatively more intense in the TURKOVAC vaccine arm, it was thought to be related to the amount of aluminium, which is the adjuvant of the vaccine, due to the fact that none of these adverse effects had an impact on daily life and their magnitude was in a narrow area. In this context, no findings threatening safety have been encountered in either group. As a result, both vaccines administered led to an increase in antibody titres with an acceptable safety profile.
"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
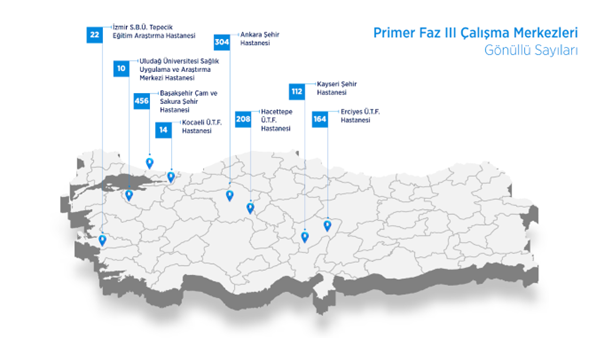
The Phase 3 study, in which the efficacy and safety evaluation of the TURKOVAC vaccine was conducted, was carried out in 8 (eight) centres in our country.

"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
The Phase 3 study, in which the efficacy and safety evaluation of the TURKOVAC vaccine was conducted, began on June 18, 2021, with the approval of the Hacettepe University Faculty of Medicine Ethics Committee. The last volunteer was included in the study on January 07, 2022.

"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
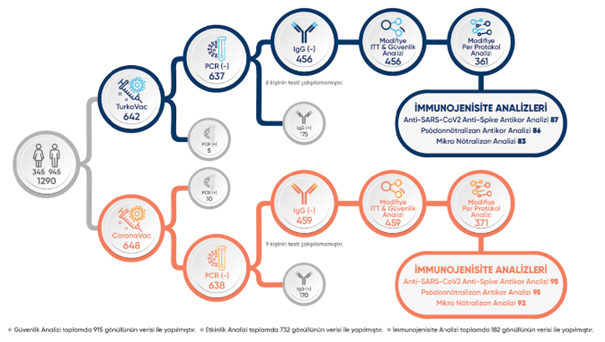
In the Phase 3 study of the TURKOVAC vaccine, 345 female and 945 male volunteers in the 18–55 age range volunteered for the study.
"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
The study process is shown as a figure.
"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
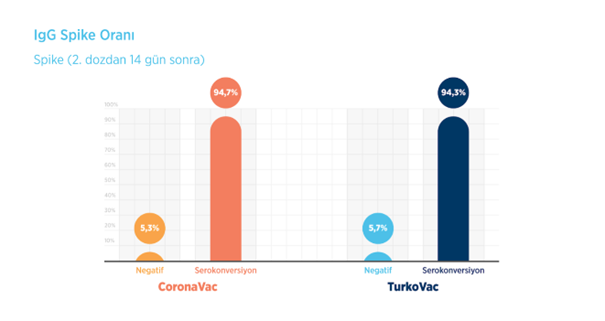
In the Phase 3 study of the TURKOVAC vaccine, antibody formation occurred in 94.7% of the volunteers in the CoronaVac arm and 94.3% of the volunteers in the TURKOVAC arm 14 days after the second dose of the vaccines.
"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
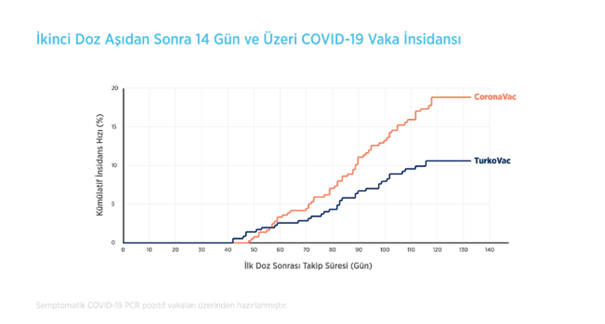
Study name- When the volunteers were followed up starting from 14 days after the second dose of TURKOVAC and CoronaVac vaccines, and the appearance of cases whose COVID-19 diagnosis was confirmed by PCR (+) among volunteers who developed COVID-19 related symptoms and complaints was evaluated according to post-vaccination days, it was observed that the cumulative number of cases was 35 in the TURKOVAC vaccine arm, while it was 61 in the CoronaVac vaccine arm within the 130-day follow-up period, as shown in the graph.

"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
Adverse effects reported by the volunteers are shown.
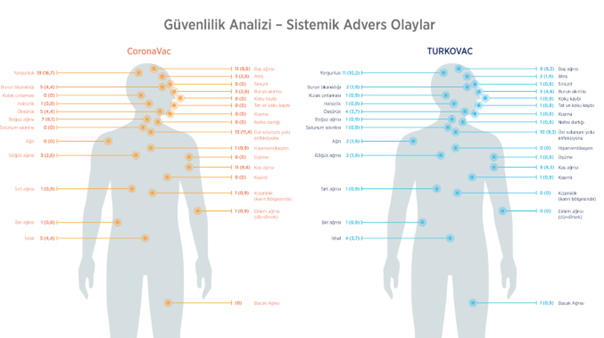
"A Randomized, Observer-Blind Phase III Clinical Study Evaluating the Efficacy, Immunogenicity, and Safety of Two Doses of Inactivated COVID-19 Vaccine (TURKOVAC) Against Two Doses of CoronaVac (Sinovac) Vaccine in Healthy Volunteers"
TSB-VAC-COV-TUR-F3.01
Adverse effects reported by the volunteers are shown.













